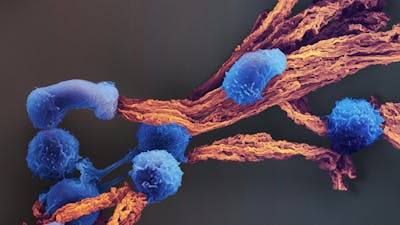
Immunotherapy, or tweaking the body’s own immune system to treat disease, is attracting significant attention in the medical field for its potential to offer long-lasting cures with fewer side effects than chemotherapy or other drugs. One type of immunotherapy involves isolating T cells (a type of white blood cell) from a patient’s body, sometimes modifying them so that they will recognize a diseased cell of interest, and rapidly producing more of them (a process called expansion). The expanded T cells are then reintroduced into the patient, where they can more effectively destroy their targets. However, current methods for modifying and expanding T cell populations are not very efficient: it can take up to a month – time that very sick patients sometimes do not have – to generate enough targeted T cells for a therapeutically effective dose.
The Wyss Institute has created a novel system for generating T cells using scaffolds that mimic the way that antigen-presenting cells (APCs) activate T cells in the body. The scaffold is comprised of biodegradable micro-rods loaded with interleukin 2 (though other cytokines can also be used) and coated with a supported lipid bilayer. This membrane can be customized with any number of T cell-activating molecules to selectively expand T cells with higher efficiency and more control than standard approaches.
We are excited about the potential of this system to tune and generate T cell products with precision that is currently not possible. Clinically, it could result in safer and more potent T cell therapies for patients who need them.
Following a single stimulation by the APC-mimetic scaffolds (APC-ms), human T cell populations displayed substantially greater expansion than current standard methods, without any loss in functionality. When tested with chimeric antigen receptor-expressing T cells (CAR-T cells) targeting lymphoma in a mouse model, the scaffolds supported greater T cell expansion and robust efficacy compared to the current standard.
This technology has been licensed.